1. Introduction
The GIMA Disposable Skin Stapler, Model 25891, is a sterile, single-use medical device designed for efficient skin suturing. It is equipped with 35 high-quality 317 L stainless steel staples. This device is intended for use by qualified medical personnel only in appropriate clinical settings such as operating rooms, critical care units, emergency departments, clinics, hospitals, and medical practices.
The design of this skin stapler aims to reduce suturing time, minimize the risk of infection, promote less tissue reaction, result in thinner scars, and facilitate faster wound healing. Its operation is designed for ease of use and provides a practical solution for skin closure.
2. Safety Information
- For Professional Use Only: This medical product must only be used by trained and qualified medical personnel.
- Sterile Product: The device is sterilized with ethylene oxide. Do not use if the sterile packaging is compromised or damaged.
- Single-Use Device: This skin stapler is designed for single use on a single patient. Do not re-sterilize or reuse. Reuse may lead to infection or device malfunction.
- Proper Disposal: Dispose of the device and all associated waste according to local regulations for medical waste.
- Storage: Store in a cool, dry place, away from direct sunlight and extreme temperatures.
- Patient Assessment: Always assess the patient's skin condition and wound characteristics to determine the suitability of skin stapling.
3. Product Components
Each GIMA Disposable Skin Stapler unit includes:
- One (1) GIMA Disposable Skin Stapler device.
- Thirty-five (35) high-quality 317 L stainless steel staples pre-loaded within the device.

Image 3.1: The GIMA Disposable Skin Stapler, pre-loaded with 35 staples made of high-quality 317 L stainless steel, designed for medical use.
4. Setup
Prior to use, ensure the following:
- Verify the integrity of the sterile packaging. Do not use if the package is opened or damaged.
- Confirm the expiration date on the packaging. Do not use expired products.
- Ensure the patient's skin is properly prepared according to standard surgical protocols (e.g., cleaned and disinfected).
- Position the patient appropriately to allow clear access to the incision site.
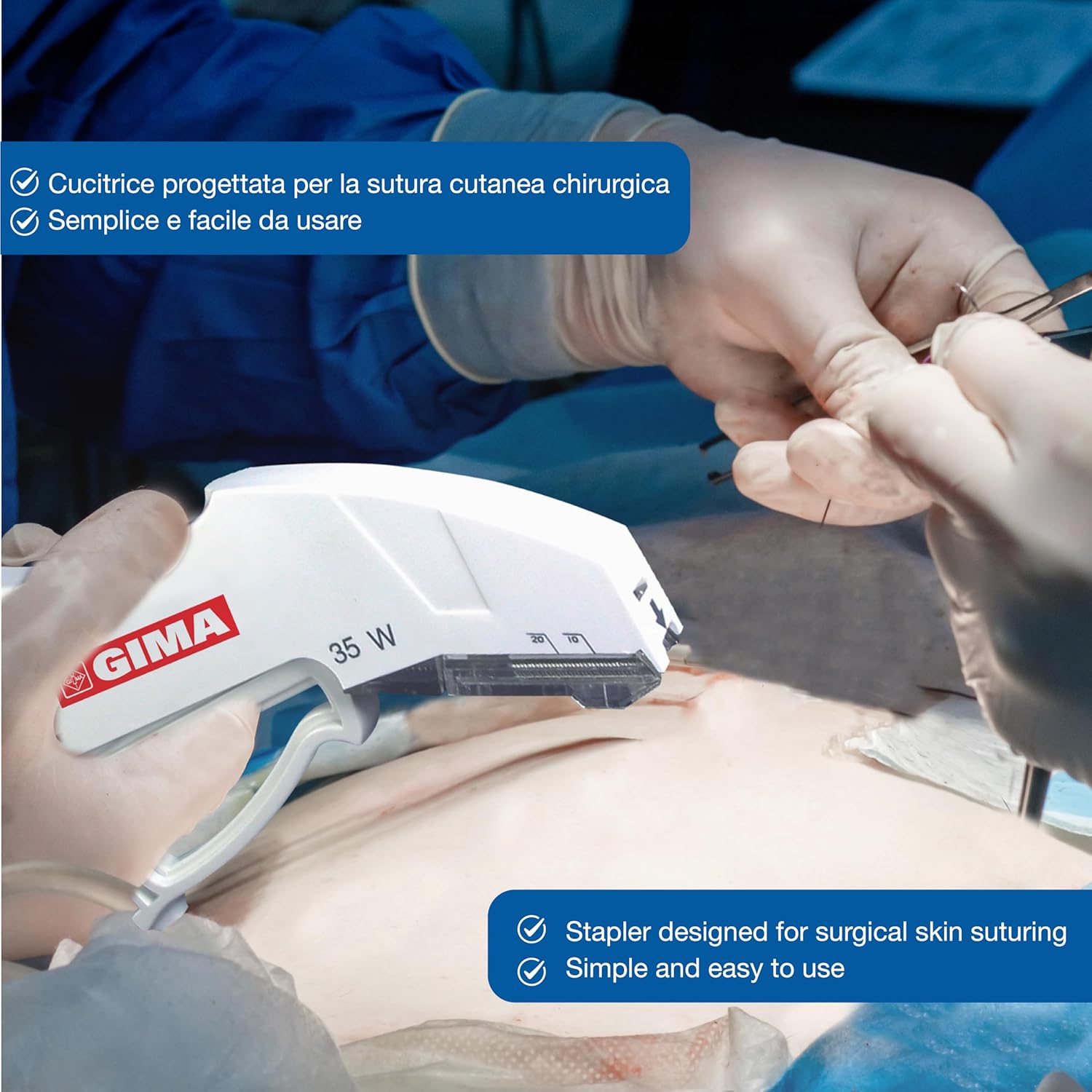
Image 4.1: A medical professional using the GIMA skin stapler for surgical skin suturing, demonstrating its simple and easy application.
5. Operating Instructions
Follow these steps for proper application of the GIMA Disposable Skin Stapler:
- Positioning the Stapler: Ensure that the front part of the skin stapler is properly positioned over the wound. The stapler should be held vertically along the longitudinal axis of the incision.
- Aligning with Incision: Gently place the skin stapler onto the incision until the front arrow on the device is aligned with the midline of the incision.
- Applying the Staple: Press the handle of the stapler firmly and smoothly to deploy a staple. Ensure the staple fully penetrates the skin edges and forms a proper closure.
- Repeat as Necessary: Continue applying staples along the incision, maintaining appropriate spacing between each staple, until the wound is adequately closed.
- Post-Application: Gently press on the applied staples to facilitate subsequent staple removal, if needed.

Image 5.1: Step-by-step diagram illustrating the correct procedure for using the GIMA skin stapler, from positioning to staple application.
Benefits of Use:
- Reduced Suturing Time: The high stapling speed helps shorten the overall suturing time.
- Lower Infection Risk: Efficient closure contributes to a reduced risk of cross-infections.
- Improved Cosmetic Outcome: Leads to less tissue reaction and thinner scars after operations.
- Faster Healing: Promotes quicker wound healing.
- Painless Removal: Staples can be removed painlessly and conveniently using a special staple remover (sold separately).

Image 5.2: A medical professional, illustrating the efficiency and reduced suturing time achieved with the GIMA skin stapler, which helps avoid cross-infections.

Image 5.3: This image represents the benefits of thinner scars and faster wound healing, which are outcomes associated with the use of the GIMA skin stapler.

Image 5.4: A healed incision, demonstrating the painless and convenient removal of staples, which is facilitated by a special staple remover.
6. Maintenance & Disposal
As the GIMA Disposable Skin Stapler is a single-use, sterile device, no maintenance is required. After a single use on a single patient, the device must be disposed of immediately.
- Disposal: Place the used skin stapler in an approved sharps container or biohazard waste receptacle.
- Compliance: Ensure disposal procedures comply with all local, national, and institutional regulations for medical and hazardous waste.
7. Troubleshooting
Given that the GIMA Disposable Skin Stapler is a sterile, single-use device, troubleshooting is generally not applicable. If the device appears to be malfunctioning or damaged prior to use, do not use it. Dispose of the faulty device safely and use a new, intact sterile unit.
8. Specifications
| Feature | Detail |
|---|---|
| Brand | GIMA |
| Model Number | 25891 |
| Staple Material | High-quality 317 L Stainless Steel |
| Staple Count | 35 staples per device |
| Sterilization Method | Ethylene Oxide |
| Usage | Single-use, disposable |
| Manufacturer | Advan |
| Package Dimensions | 25.4 x 17.7 x 12.4 cm |
| Item Weight | 300 g |

Image 8.1: The GIMA skin stapler shown with its approximate dimensions, indicating a height of 6.4 inches (16 cm).
9. Warranty and Support
Information regarding specific warranty terms or direct customer support for the GIMA Disposable Skin Stapler is not provided in this documentation. For any inquiries, please contact your medical device supplier or GIMA's official distribution channels.





