Montaz B088CCV3DP
Montaz 125 - Vial of 1 gm Injection User Manual
Comprehensive Guide for Safe and Effective Use
Product Overview
The Montaz 125 is a sterile vial containing a combination of Ceftriaxone Sodium IP equivalent to Ceftriaxone 1 g and Tazobactam 125 mg. This product is intended for intravenous (I.V.) or intramuscular (I.M.) use only, as indicated on the packaging. It is manufactured by Aristo Pharmaceuticals Pvt. Ltd. and is designed for specific medical applications.
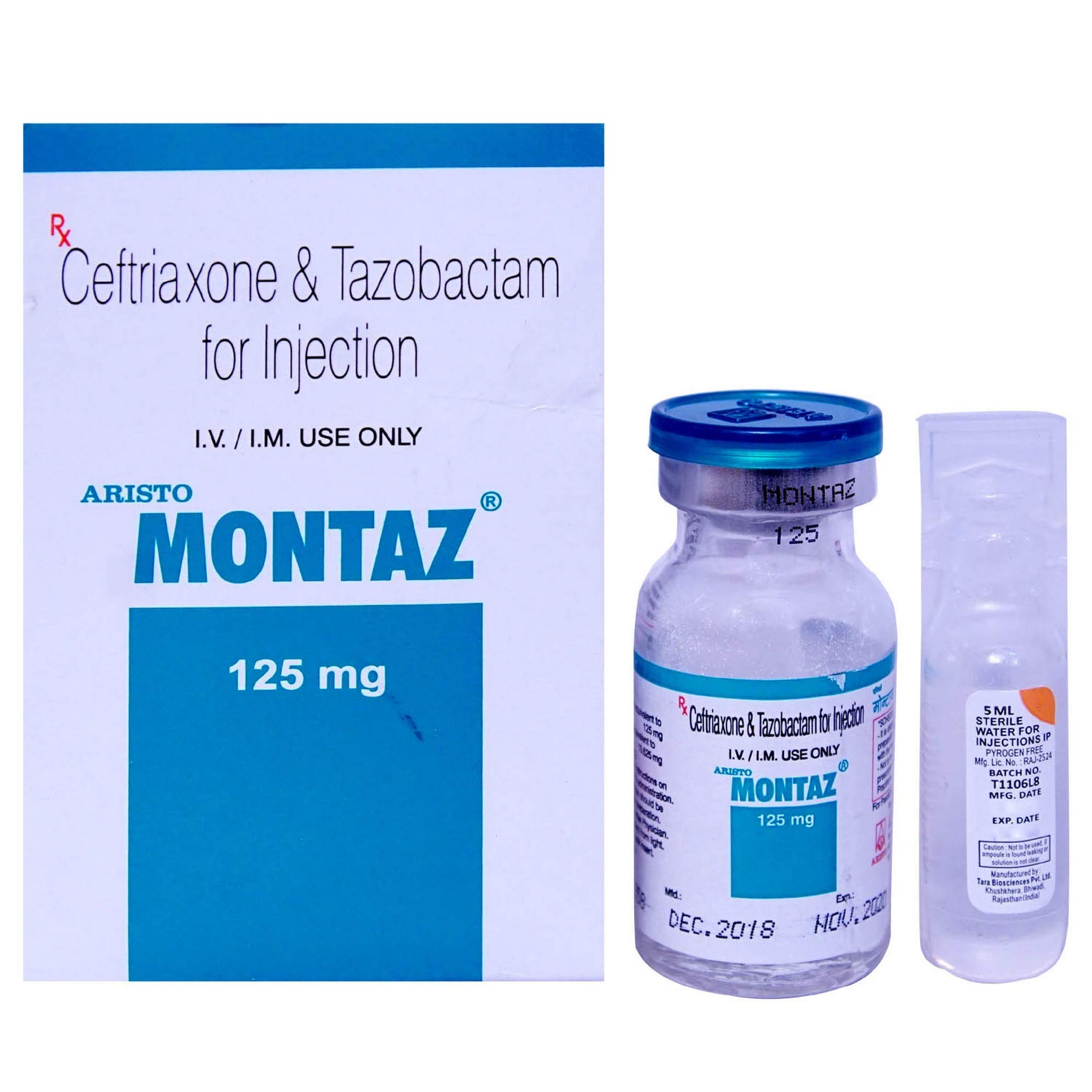
Image: Montaz 125 vial and its outer packaging. The packaging clearly states "Ceftriaxone & Tazobactam for Injection" and "I.V. / I.M. USE ONLY". The vial itself is labeled "MONTAZ 125 mg" and shows manufacturing and expiry dates. A separate smaller vial of sterile water for injections is also visible.
Specifications
| Manufacturer | Aristo Pharmaceuticals Pvt. Ltd. |
| Composition | Ceftriaxone Sodium IP equivalent to Ceftriaxone 1 g, Tazobactam 125 mg |
| Item Part Number | B088CCV3DP |
| Product Dimensions | 14 x 4 x 2 cm |
| Item Weight | 1 g |
| Net Quantity | 1.00 Count |
| Date First Available | 8 May 2020 |
Setup and Preparation
This product is a prescription medication and must be prepared and administered by a qualified healthcare professional.
- Verify Product Integrity: Before use, carefully inspect the vial for any signs of damage, cracks, or leakage. Ensure the solution inside is clear and free from particulate matter. Do not use if the ampoule is found leaking or the solution is not clear.
- Check Expiry Date: Confirm that the manufacturing and expiry dates printed on the vial and packaging are valid. Do not use expired medication.
- Reconstitution: Montaz 125 requires reconstitution with a suitable diluent, typically sterile water for injections. The specific volume and method of reconstitution should follow standard medical protocols and the instructions provided by the prescribing physician or pharmacist.
- Aseptic Technique: Always use strict aseptic technique during preparation and administration to prevent contamination.
Operating and Administration
Montaz 125 is for intravenous (I.V.) or intramuscular (I.M.) use only. Administration should be performed by a trained medical professional.
- Dosage: The dosage and duration of treatment will be determined by the prescribing physician based on the patient's condition, age, weight, and response to therapy.
- Administration Route:
- Intravenous (I.V.) Administration: Administer slowly over the recommended period as directed by the physician.
- Intramuscular (I.M.) Administration: Administer deeply into a large muscle mass.
- Monitoring: Patients should be monitored for adverse reactions during and after administration.
- Compatibility: Ensure compatibility with other medications or solutions if co-administration is required. Consult a pharmacist or physician for guidance.
Storage and Maintenance
Proper storage is crucial to maintain the efficacy and safety of Montaz 125.
- Unreconstituted Vial: Store the unopened vial at room temperature, away from direct sunlight and moisture, as per manufacturer's recommendations. Do not freeze.
- Reconstituted Solution: Once reconstituted, the solution should be used immediately. If not used immediately, follow specific storage guidelines provided by the manufacturer or pharmacist regarding stability and refrigeration. Discard any unused portion after the recommended time.
- Disposal: Dispose of all used vials, syringes, and needles according to local medical waste disposal regulations. Do not dispose of in household trash.
Troubleshooting and Precautions
This section provides general guidance. Always consult a healthcare professional for specific medical advice.
- Adverse Reactions: If any unexpected or severe reactions occur during or after administration, discontinue use immediately and seek urgent medical attention.
- Allergic Reactions: Be aware of potential allergic reactions. Symptoms may include rash, itching, swelling, severe dizziness, or trouble breathing.
- Incomplete Dissolution: If the powder does not dissolve completely upon reconstitution, do not use the solution.
- Contamination: If contamination is suspected during preparation or administration, discard the product and prepare a new dose using sterile technique.
Legal Disclaimer: Manufacture, Expiry dates and other information are mentioned on the products. Please always read labels and warnings before using or consuming the product. This product is a prescription medication and should only be used under the guidance of a registered medical practitioner.
Warranty and Support
As a pharmaceutical product, Montaz 125 does not typically come with a consumer warranty in the traditional sense. Its quality and efficacy are guaranteed by the manufacturer, Aristo Pharmaceuticals Pvt. Ltd., provided it is stored and used according to instructions.
For any product-related inquiries, concerns, or to report adverse events, please contact your prescribing physician, pharmacist, or the manufacturer directly.
- Manufacturer: Aristo Pharmaceuticals Pvt. Ltd.
- Product ASIN: B088CCV3DP
- Returns Policy: Please refer to the purchasing platform's returns policy for pharmaceutical products. Due to the nature of the product, returns may be restricted.
Ask a question about this manual
Ask about setup, troubleshooting, compatibility, parts, safety, or missing instructions. Manuals+ will review the question and use this page’s manual context to help answer it.